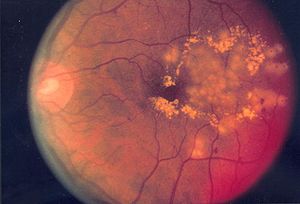
There is no neat, targeted way to treat diabetic retinopathy, a condition that could lead to blindness. Laser therapy can result in diminished side and night vision and the other current method used, the cancer drug docetaxel, clear from the system so quickly that high doses are needed, increasing toxicity to healthy tissue. The research group ANPRON tells us about a team of Canadian scientists who think they have found a solution for sufferers of diabetic retinopathy. They have made a MEMS device (micron-sized electromechanical systems) that could be implanted behind the eye and release docetaxel on command by an external magnet.
People from BioMEMS research group @ University of auckland stated that
Their most recent work (Wlodkowic D, Khoshmanesh K, Sharpe JC, Darzynkiewicz Z, Cooper JM. Apoptosis goes on a chip: advances in the microfluidic analysis of programmed cell death. Anal Chem. 2011 Jun 16. [Epub ahead of print]) provides an innovative summary of the recent advances in miniaturized chip-based devices for the analysis of programmed cell death.it provides future prospects of the Lab-on-a-Chip devices with wide reaching perspectives in anti-cancer drug discovery and high-throughput cell-based screening routines.
Abstract:
Soon, drug delivery that precisely targets cancerous cells without exposing the healthy surrounding tissue to the medication’s toxic effects will no longer be an oncologist’s dream but a medical reality, thanks to the work of Professor Sylvain Martel, Director of the Nanorobotics Laboratory at Polytechnique Montréal.

Known for being the world’s first researcher to have guided a magnetic sphere through a living artery, Professor Martel is announcing a new breakthrough in the field of nanomedicine. Using a magnetic resonance imaging (MRI) system, his team successfully guided
microcarriers loaded with a dose of
anti-cancer drug through the bloodstream of a living rabbit, right up to a targeted area in the liver, where the drug was successfully administered. This is a medical first that will help improve chemoembolization, a current treatment for liver cancer.
Microcarriers on a mission
This is a preview of World First: Localized Delivery of an Anti-Cancer Drug by Remote-Controlled Microcarriers.
Read the full post (349 words, 2 images, estimated 1:24 mins reading time)

Cancer cells photographed by camera attached to microscope in time-lapse manner. Image via Wikipedia
The upside of chemotherapy is that it attacks cancer cells and kills them. The downside – and a steep downside it is – is that it is composed of highly toxic compounds that attack other cells of the body, too, resulting in any number of harmful side effects, from anemia to hair loss to nausea and vomiting.
The question concerning researchers is how do we deliver chemotherapy drugs to the harmful cells and leave the healthy cells alone?
This is a preview of Innovation in Targeted Drug Delivery Using Gold Coatings.
Read the full post (509 words, 1 image, estimated 2:02 mins reading time)