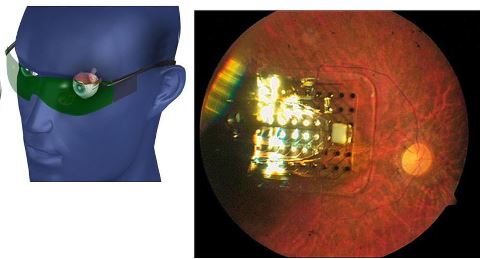
FDA reviewers expressed concern over the long-term safety of what could be the first retinal prosthesis device despite evidence that it helps improve the vision of some nearly blind patients.
Members of the FDA’s Medical Devices Advisory Committee will vote Friday on whether to recommend approval of the Argus II Retinal Prosthesis device. Members of the ophthalmic panel also will discuss and recommend possible post-approval study requirements should the device ultimately gain FDA clearance.
The Argus II is designed for use by patients with severe to profound retinitis pigmentosa, who experience progressive vision loss, often leading to blindness.

Medical bionics is the replacement or monitoring of damaged organs through engineered devices that interface with the body to improve health outcomes. In this presentation I will concentrate on medical bionic devices designed to restore or supplement function of the
nervous system lost during disease or injury.
A number of commercially available neural prostheses will be described – including the remarkably successful bionic ear and deep brain stimulation for movement control.
I will then review some of the current research performed around the world – including recent developments in brain-machine interface that will ultimately allow patients to control prosthetic limbs and wheel chairs; developments in functional electrical stimulation for gait and standing in paraplegia; and research to develop a prosthetic balance system.